
Human Tissue Research Services
We offer access to a large bank of over 550,000 Japanese clinical specimens annotated with donor demographic data and information from medical records. Our bank is drawn from a network of leading medical institutions in Japan, and is available to drug discovery and life science researchers via our portfolio of contract research services.
Tissue Collection
Our human biospecimen bank is comprised of FFPE (formalin-fixed paraffin embedded) and fresh frozen tissues collected during the course of medical treatment. Its characteristics include:
- Access to pathology reports and donor medical records
- Encompasses specimens from the oncology, pulmonary, CNS, cardiovascular and other disease areas
- True normal (autopsy-derived) material is also available
- All tissues are QCed by certified histopathologists
- Use of all tissues is ethically approved
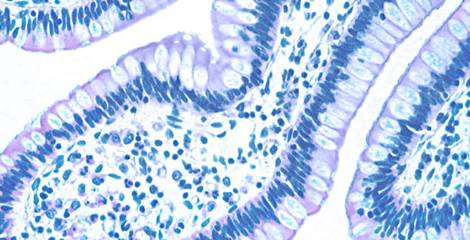
Analytical Services
Our team has extensive experience of drug discovery research studies utilizing human biospecimens, and is committed to delivering scientific data of the highest quality. We can analyze biospecimens using a variety of methodologies:
- Single stain IHC (immunohistochemistry)
- Double / triple stain & fluorescent IHC
- In situ hybridization (ISH, FISH)
- DNA Microarray (GeneChip etc.)
- Others -please inquire
IHC Studies
IHC on client targets with specialized pathology interpretation is a core expertise. Study set-up is flexible and designed to unequivocally answer the scientific questions posed. The table below outlines some key features of our IHC studies:
| Aspect | Notes |
| Antibodies | Specificity of client or commercial antibodies is validated using control tissues and / or cell lines. |
| Tissues | We usually provide the tissues but client-provided tissues can also be analyzed. |
| Staining | Carried out in clinically-certified facilities with automated staining to ensure reproducibility. Client specific protocols can be used. |
| Interpretation | Carried out by certified pathologists with extensive experience of human tissue IHC in a drug discovery context. |
| Reporting | Reporting is tailored to your requirements. |
Why Japanese Biospecimens?
Human biomaterials are a crucial resource for modern drug discovery research and development. One key application is in the study of inter-individual biological variations that may significantly impact the relevance of drug targets and biomarkers, as well as the behavior of drugs in the body. Much of this biological variation arises from ethnic differences, as well as lifestyle and environmental factors.
Therefore, in studies utilizing human biomaterials, it makes sense from both a scientific and business perspective to include biomaterials from donors representing the main target markets.
Valued at ~80 billion USD (2019), Japan is the world’s second largest pharmaceutical market. If Japan is a potential market for your drug, it is thus highly advisable to create data using biomaterials from a Japanese donor population.
- Gain insight into potential biological differences to better inform scientific and business decisions
- For regulatory filings, the Japanese regulatory authorities favorably regard Japanese biomaterial-derived data as being more predictive
However, it can be extremely difficult to access Japanese clinical biospecimens for commercial research due to cultural, bureaucratic and other hurdles, especially for non-Japanese companies. This is where access to our unique biobank can assist your research program.
Working With Us

An experienced bilingual manager is assigned to your project to provide you with a single point of contact and robust project management.
Your study plan is custom-created based on a thorough understanding of your objectives as well as budget and timeline needs. Importantly, we ensure the study design accounts for any differences in pathology practices between Japan and other regions. Ongoing study updates and interim reports are provided as required.
Further Information
If you have any questions or require more information, please do not hesitate to contact us. We would be pleased to discuss how we can help you achieve your research objectives.
